How we discovered the structure of the atom.
The history of the atom is a long one which underwent several theories, changes and controversies. You might have heard of the Greek philosopher Democritus who, while sitting by the beach and observing the sand, posed the theory that everything was made of indestructible building blocks he called atoms (the word atomos means indivisible.) In the 19th century, John Dalton was the first scientist to discover the atom by means of experimentation, and later that century, J.J. Thomson discovered that atoms contained electrons, showing that they have an internal structure and are not, in fact, the uniformly spherical indivisible particles they were thought to be.

J.J. Thomson’s model of the atom was known as the ‘plum pudding model’ because it assumed that atoms consisted of positive charge which is uniformly distributed, and electrons scattered between the positive charges (just like raisins in a plum pudding.)
Of course, this is not the model of the atom we are used to today. The way the current model of the atom was discovered was through an experiment conducted by the physicist Ernst Rutherford and his colleagues in 1909: the Rutherford Scattering Experiment.
Scientists in Rutherford’s time knew of certain things:
- They knew that particles called alpha particles existed, which were quite heavy and positively charged. (We now know that an alpha particle is a helium nucleus: two protons and two neutrons)
- They knew an atom contained positive and negative charges, and they assumed these were distributed in the ‘plum pudding’ manner.
- Their prediction was that if they shot an alpha particle through an atom, it would pass right through.
To test this, they set up a sheet of very very thin gold foil, and a source which shot alpha particles at it. The experiment took place in a vacuum tube and a detector was placed so that it could detect particles coming out of the foil from all directions. The initial expectation was that all the alpha particles should pass through the gold foil and be detected on the other side… and this was the case, for most of them. However, many particles were deflected, and some were even repelled straight in the opposite direction!
This was groundbreaking. The only way the alpha particle could be completely repelled was if all the mass of the atom was concentrated in a very small part, and all the positive charge was concentrated too. That would also explain why some of the particles were deflected: they did not hit the nucleus but were close to it and were therefore repelled by it.
The Rutherford scattering experiment led to the model of the atom we have today, one where:
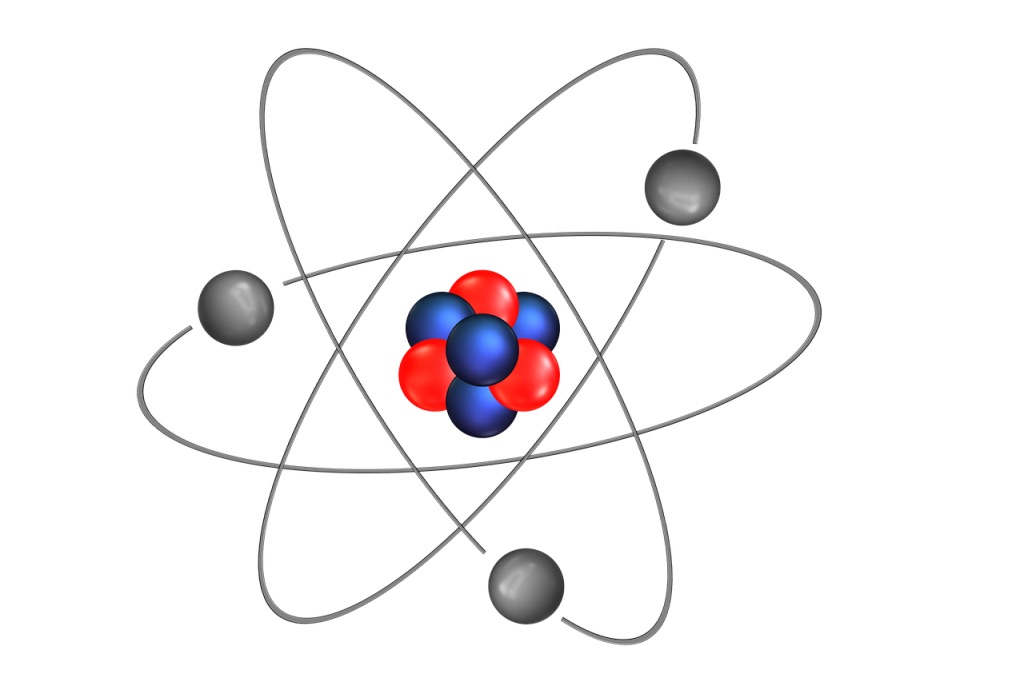
1) Most of the mass of the atom comes from the nucleus.
2) The nucleus is positively charged.
3) Most of the atom consists of empty space (which is why most alpha particles passed through with no change in direction)
We’ve come a long way since Democritus, I think that’s safe to say…
Leave a comment