It seems intuitive that a solid of a particular substance would sink when placed inside the liquid form of that substance (a solid ball of argon will sink in a bowl of liquid argon.) And yet, we have icebergs, ice sheets to skate on, and ice cubes floating at the top of our lemonades. Despite what our intuition says, ice floats on water… why?
First, we have to question where our intuition is coming from. The reason we assume solid sinks in liquid (of the same substance) is because this is the model we are used to:
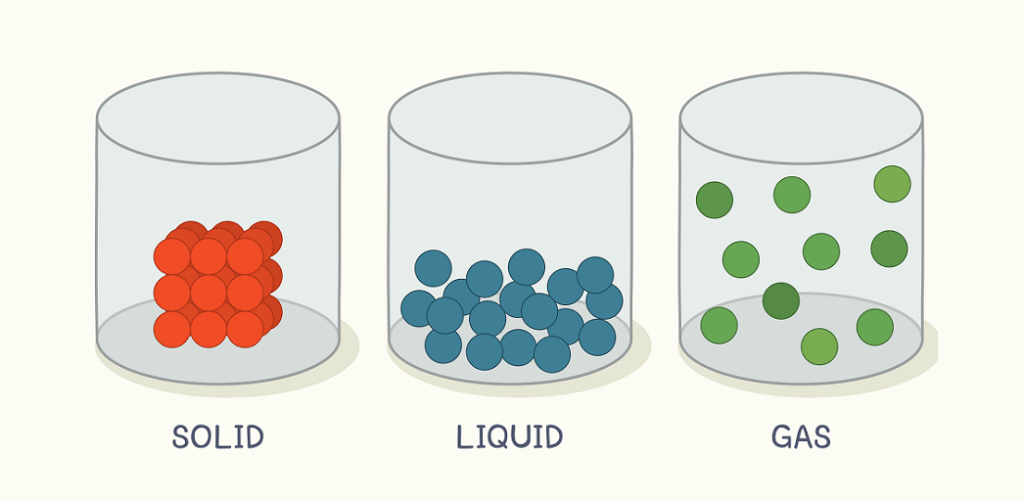
…Where it is clear that the solid particles are more tightly and regularly arranged, causing it to have a higher density than the liquid. Objects will sink if they have a higher density than the liquid they’re placed in.
So, how come water doesn’t follow this rule? Can it be that ice is less dense than water?
The answer: yes, it is. And this is because of a phenomenon called hydrogen bonding.
To understand hydrogen bonding, we have to rewind a little and look at the concept of electronegativity. Electronegativity is the ability of an atom to attract electrons to itself. Not all atoms attract electrons equally; some do so far more strongly than others. The top most electronegative elements are fluorine, chlorine, oxygen and nitrogen. In compounds containing one of these elements—such as in water, H2O—the electron pair will be closer to the oxygen nucleus than the hydrogen nucleus. This means the oxygen atoms will be slightly more negatively charged, and the hydrogen slightly more positive.
We all know the rule: opposite charges attract (a fact that cliche romance movies love to take advantage of.) So what happens is that the positive hydrogen atom of one molecule is attracted to the negative oxygen atom of the other. This intermolecular force of attraction is called hydrogen bonding.
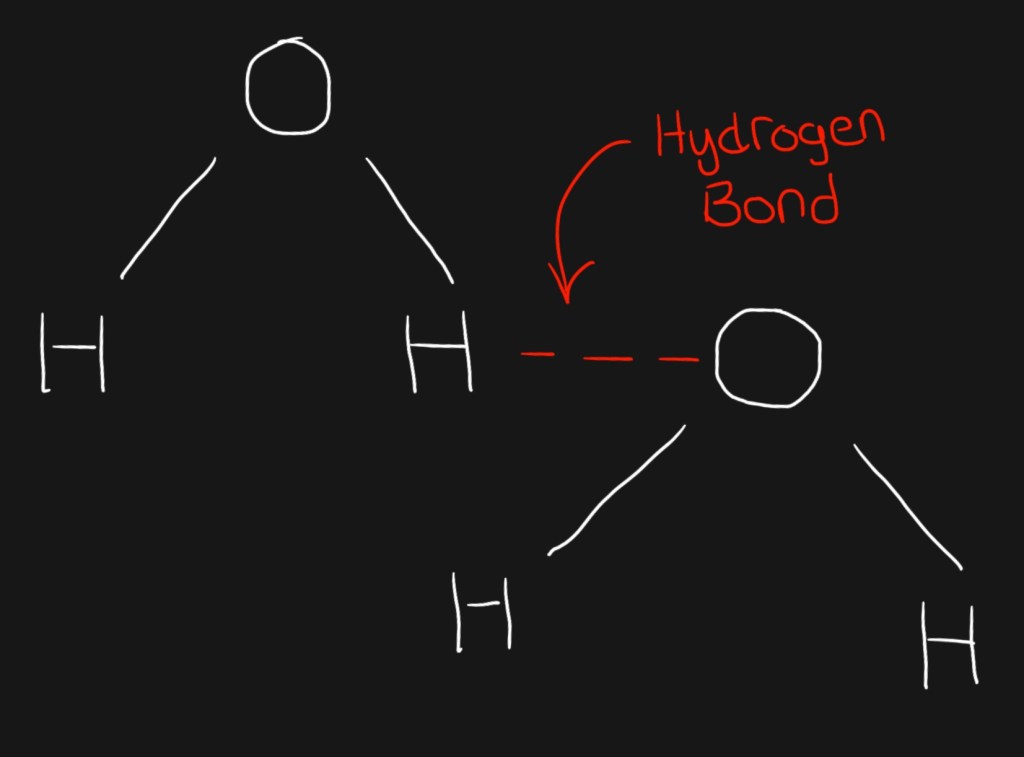
You might be wondering what this has to do with ice floating in water… it seems an awful lot of information for such a simple question. Well, you see, the hydrogen bonds are strongest when water is in solid form, and the arrangement they form looks like this:
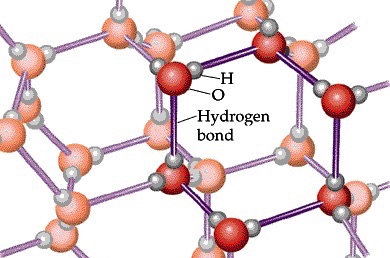
A beautiful crystalline pattern. Because of this arrangement, the water molecules are further apart in solid form than they are in liquid. In liquid form, the hydrogen bonds form and break quite easily, meaning the particles don’t end up being regularly spaced as in ice and tend to come closer together.
The reason why the sea doesn’t freeze over, the reason why we have ice skating rinks and, simply put, the reason why the titanic sunk, essentially comes down to the structure of the H2O molecule.
And this is the beauty of nature… something so small and simple as the position of a pair of electrons, causes such significant effects as to shape the world as we know it.
Leave a comment